Insecticide
Alpha-cypermethrin
TC & FORMULATION: 95%TC, 5% EC, 10%EC, 10%SC
Structure Formula: 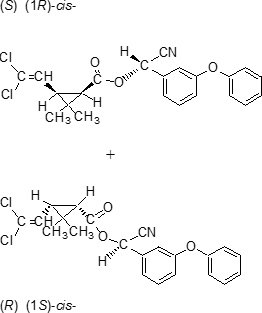
NOMENCLATURE
Common name alpha-cypermethrin (BSI, draft E-ISO); alpha-cyperméthrine ((f) draft F-ISO)
IUPAC name A racemate comprising (S)-a-cyano-3-phenoxybenzyl (1R,3R)-3-(2,2-dichlorovinyl)-2,2-dimethylcyclopropanecarboxylate and (R)-a-cyano-3-phenoxybenzyl (1S,3S)-3-(2,2-dichlorovinyl)-2,2-dimethylcyclopropanecarboxylate
Roth: A racemate comprising (S)-a-cyano-3-phenoxybenzyl (1R)-cis-3-(2,2-dichlorovinyl)-2,2-dimethylcyclopropanecarboxylate and (R)-a-cyano-3-phenoxybenzyl (1S)-cis-3-(2,2-dichlorovinyl)-2,2-dimethylcyclopropanecarboxylate
Chemical Abstracts name [1a(S*),3a]-(?-cyano(3-phenoxyphenyl)methyl 3-(2,2-dichloroethenyl)-2,2-dimethylcyclopropanecarboxylate
Other names alfoxylate*; alphamethrin*(rejected common name proposals)
CAS RN [67375-30-8] correct stereochemistry; [52315-07-8] (formerly [86752-99-0], [86753-92-6]) cypermethrin (no stereochemistry stated) were sometimes used in Chemical Abstracts
PHYSICAL CHEMISTRY
Composition Tech. grade alpha-cypermethrin is >90% pure m/m, typically >95%. Mol. wt. 416.3 M.f. C22H19Cl2NO3 Form Colourless crystals; (tech. is a white to pale powder, with a weak aromatic odour). M.p. 81.5 ºC (97.3 %) B.p. 200 ºC/9.3 Pa V.p. 2.3 ´ 10-2 mPa (20 ºC) KOW logP = 6.94 (pH 7) Henry 6.9 ´ 10-2 Pa m3 mol-1 (calc.) S.g./density 1.28 (22 ºC) Solubility In water 0.67 (pH 4), 3.97 (pH 7), 4.54 (pH 9), 1.25 (double distilled water) (all in µg/l, 20 °C). In n-hexane 6.5, toluene 596, methanol 21.3, isopropanol 9.6, ethyl acetate 584, acetone:hexane >0.5 (all in g/l, 21 °C); miscible in dichloromethane and in acetone (>10?g/l). Stability Very stable in neutral and acidic media, hydrolysed in strongly alkaline media; DT50 (pH 4, 50 °C) stable over 10 d, (pH 7, 20 °C) 101 d, (pH 9, 20 °C) 7.3 d. Thermally stable up to 220 ºC. Field data indicate that, in practice, it is stable to air. F.p. >80 ºC (closed cup); not highly flammable
APPLICATIONS
Biochemistry Acts by preventing transmission of impulses along nerves, brought about by blocking the passage of sodium ions through sodium channels in nerve membranes thus preventing action potentials passing down axons. Typically this intoxication results in a rapid "knockdown" and resultant mortality. Mode of action Non-systemic insecticide with contact and stomach action. Acts on the central and peripheral nervous system in very low doses. Uses Control of a wide range of chewing and sucking insects (particularly Lepidoptera, Coleoptera, and Hemiptera) in fruit (including citrus), vegetables, vines, cereals, maize, beet, oilseed rape, potatoes, cotton, rice, soya beans, forestry, and other crops; applied at 10-15 g/ha. Control of cockroaches, mosquitoes, flies, and other insect pests in public health; and flies in animal houses. Also used as an animal ectoparasiticide. Formulation types EC; SC; TB; UL; WP. Compatibility Compatible with most organophosphorus insecticides.
About price and product information, please click Message button.
Open the consulting page, please Writing information, customer manager will reply within 24 hours!
or Contact email:Donna.wen@molotuschem.com
Cell-phone number:17717500809
Tel:0086 21 61203246
Fax:0086 21 61612025
Address:Room 402, No.553, Maotai Road, Changning District, Shanghai.