Herbicide
Clethodim
TC & FORMULATION: 90%TC, 120G/L EC, 240G/L EC
Structure Formula: 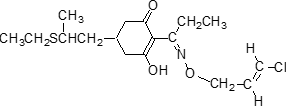
NOMENCLATURE
Common name clethodim (BSI, ANSI, draft E-ISO); clétodime ((f) draft F-ISO)
IUPAC name (?-2-[(E)-1-[(E)-3-chloroallyloxyimino]propyl]-5-[2-(ethylthio)propyl]-3-hydroxycyclohex-2-enone
Chemical Abstracts name (E,E)-(?-2-[1-[[(3-chloro-2-propenyl)oxy]imino]propyl]-5-[2-(ethylthio)propyl]-3-hydroxy-2-cyclohexen-1-one
CAS RN [99129-21-2]
PHYSICAL CHEMISTRY
Composition Tech. is >91% pure. Mol. wt. 359.9 M.f. C17H26ClNO3S Form Clear, amber liquid. B.p. Decomposes below boiling point. V.p. <1 ´ 10-2 mPa (20 ºC) S.g./density 1.14 (20 ºC) Solubility Soluble in most organic solvents. Stability Aqueous hydrolysis DT50 28 d (pH 5), 300 d (pH 7), 310 d (pH 9). Aqueous photolytic DT50 (sterile buffers, pH 5, 7 and 9) 1.7-9.6 d (without photosensitiser), 0.5-1.2 d (with photosensitiser).
APPLICATIONS
Biochemistry Fatty acid synthesis inhibitor, by inhibition of acetyl CoA carboxylase (ACCase). Mode of action Systemic herbicide, rapidly absorbed and readily translocated from treated foliage to the root system and growing parts of the plant. Uses Post-emergence control of annual and perennial grasses, at 60-240 g/ha, in a wide range of broad-leaved crops (including such field crops as soya beans, cotton, flax, sunflowers, alfalfa, peanuts, oilseed rape, sugar beet, tobacco, and potatoes), vegetable crops, trees and vines. To be used with a non-phytotoxic crop oil concentrate. Phytotoxicity Good tolerance in broad-leaved crops. Formulation types EC. Compatibility Antagonism has been observed when tank-mixed with 'Basagran' or 'Blazer'.
About price and product information, please click Message button.
Open the consulting page, please Writing information, customer manager will reply within 24 hours!
or Contact email:Donna.wen@molotuschem.com
Cell-phone number:17717500809
Tel:0086 21 61203246
Fax:0086 21 61612025
Address:Room 402, No.553, Maotai Road, Changning District, Shanghai.